Overview
Overview of Pharmaceutical Approval Process
- Approve pharmaceuticals whose safety, efficacy, and quality are identified (including pharmaceutical ingredients) through technological review and inspection for
their manufacturing and distribution.(「Pharmaceutical Affairs Act」Article 31)
- Classification of pharmaceuticals : Drug products, pharmaceutical ingredients
- Classification of drug products : New Drug, Pharmaceutical required for data submission and Generic Drug
- New Drug refers to a new material pharmaceutical whose chemical structure or original composition is totally different from a pharmaceutical previously approved in Korea or a multiple preparation containing a new material as an active drug substance, which is designated by Minister of Food and Drug Safety. (「Pharmaceutical Affairs Act」Article 2 Subparagraph 8)
- Pharmaceutical required for data submission refers to not a new drug but a pharmaceutical required for the safety and efficacy evaluation. (Regulation on Pharmaceuticals Approval, Notification and Review (MFDS Notification) Article 2 Subparagraph 8) e.g.) pharmaceuticals with new effectiveness, new composition or increase/decrease of strength of active drug substance, and pharmaceuticals with new route of administration etc.
- Generic Drug refers to a pharmaceutical which is equivalent to a new drug (reference drug) in terms of active ingredient, dosage form and strength
Data Requirements for Approval
- New Drug (「Regulation on Safety of Pharmaceuticals」(Ordinance of the Prime Ministerial) Article 9)
- (Review by Drug Evaluation Department) Safety & efficacy data, specifications & test methods, Drug Master Files (DMF), certificate of manufacturing and marketing(Imported Pharmaceutical) Data such as name and address of manufacturers of active pharmaceutical ingredients
- (Review by Other Departments) Evaluation data of conducting of Good Manufacturing Practice (GMP)
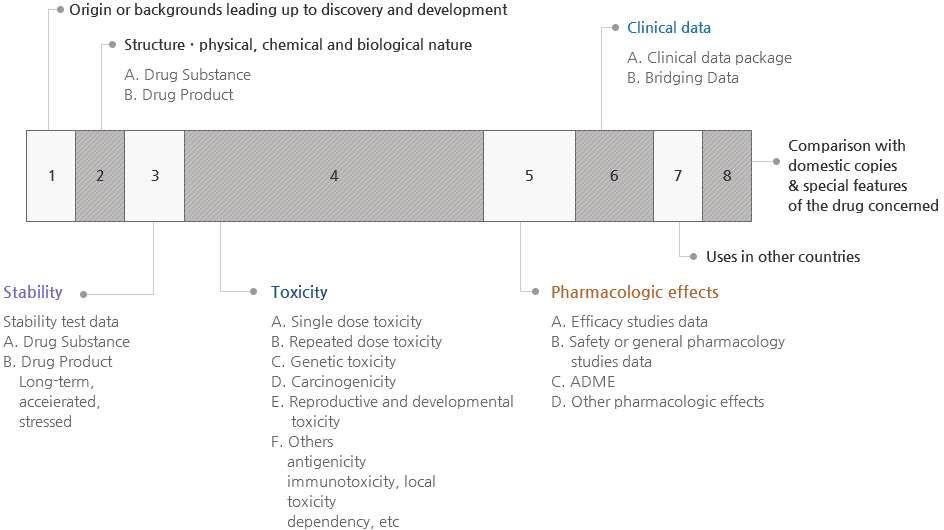
Figure 1. Dossier for Safety & Efficacy Evaluation
Data on Drug Substance
- Data on structure identification
- Data on physical & chemical properties
- Data on manufacturing methods
- Data on specifications & test methods are stated
- Supportive data on specifications & test methods
- Data on test results
- Data on reference standards, reagents, and test solutions
- Data on containers & packaging materials
Data on drug product
- Data on drug product
- Data on composition
- Data on manufacturing methods
- Data on specifications & test methods are stated
- Supportive data on specifications & test methods
- Data on test results
- Data on reference standards, reagents, and test solutions
- Data on containers & packaging materials
- Pharmaceutical required for data submission : Selectively submit data required for safety and efficacy evaluation among submission data for new drug.
- Generic Drug : Submit bioequivalence test data and quality data instead of safety and efficacy data such as toxicity, pharmacology, clinical trials. 「Regulation on
Safety of Pharmaceuticals」(Ordinance of the Prime Ministerial) Article 4 (1) 3
- Generic drugs are reviewed by Drug Evaluation Department and approved by regional FDAs.
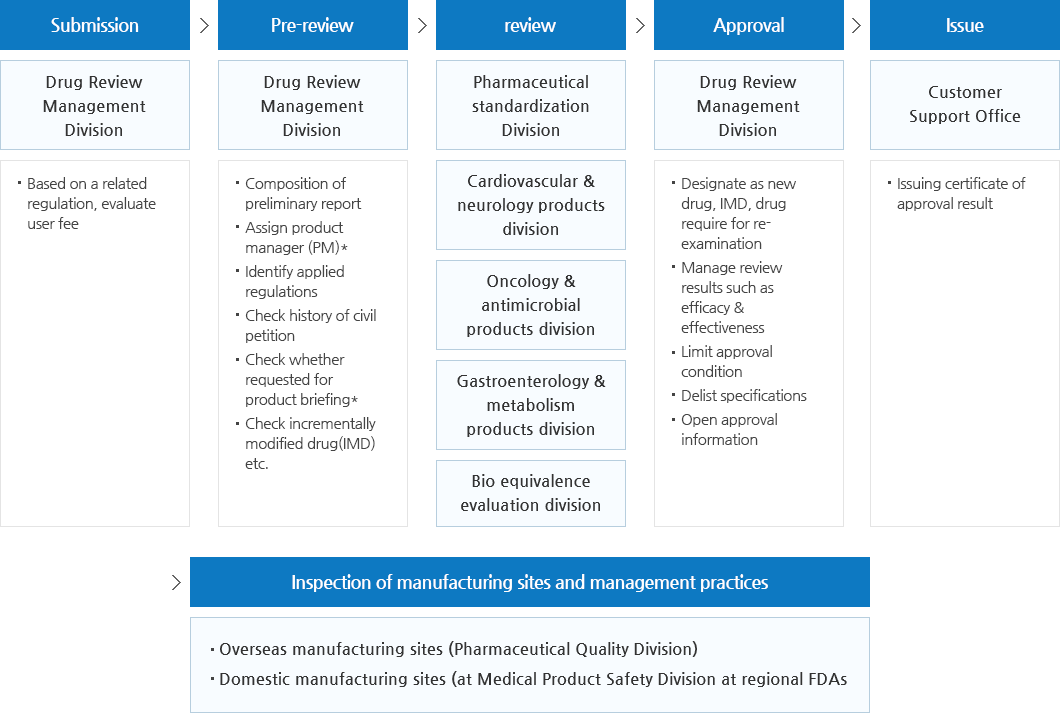
Work Flow for Approval of Pharmaceuticals
- New drug, Pharmaceutical required for data submission
 Zoom
Zoom
| Submission | Pre-review | review | Approval | Issue |
|---|---|---|---|---|
| Drug Review Management Division | Drug Review Management Division | Pharmaceeutical standardization Division | Drug Review Management Division | Customer Support Office |
| Based on related regulation, evaluate user fee | Composition of preliminary report Assign product manager (PM)* Identify applied regulations Check history of civil petition Check whether requested for product briefing* Check incrementally modified drug(IMD) etc. |
Cardiovascular & neurology products division | Designate as new drug. IMD, drug require for reexamination Manage review results such as efficacy & effectiveness Limit approval condition Delist specifications Open approval information |
Issuing certificate of approval result |
| Onclogy & antimicrobial products division | ||||
| Gastroenterology & metabolism products division | ||||
| Bio equivalence evaluation divistion |
| Inspection of manufacturing and management practices |
|---|
| Overseas manufacturing sites(Pharmaceutical Quality Division) Domestic manufacturing sites(at Medical Product Safety Division at regional FDAs) |
- In the whole process of drug review and approval, consult with Central Pharmaceutical Affairs Advisory Committee (CPAC), if necessary
- Product Manager (PM) a general manager from submission, pre-review, supplementation, approval and revision for each product
- Pre-review system : PM examines submission data and the adequacy of data requirements
- Product Briefing
- (Priority) new drug, IMD and when requested by civil petitioner
- (Participants) civil petitioner, Review Division, Pharmaceutical Policy Division
Improve efficiency and predictability of review and approval process by enhancing mutual understanding between reviewer and petitioner of a product required for approval process - Drug Approval Update
Weekly withdrawals, monthly approvals, approval report (NDA), evaluation results of safety and efficacy (pharmaceuticals required for data submission)
- Generic Drug
- Required Application Documents : Bioequivalence study, GMP documents and CMC(Chemistry, Manufacturing, Controls) data
 Zoom
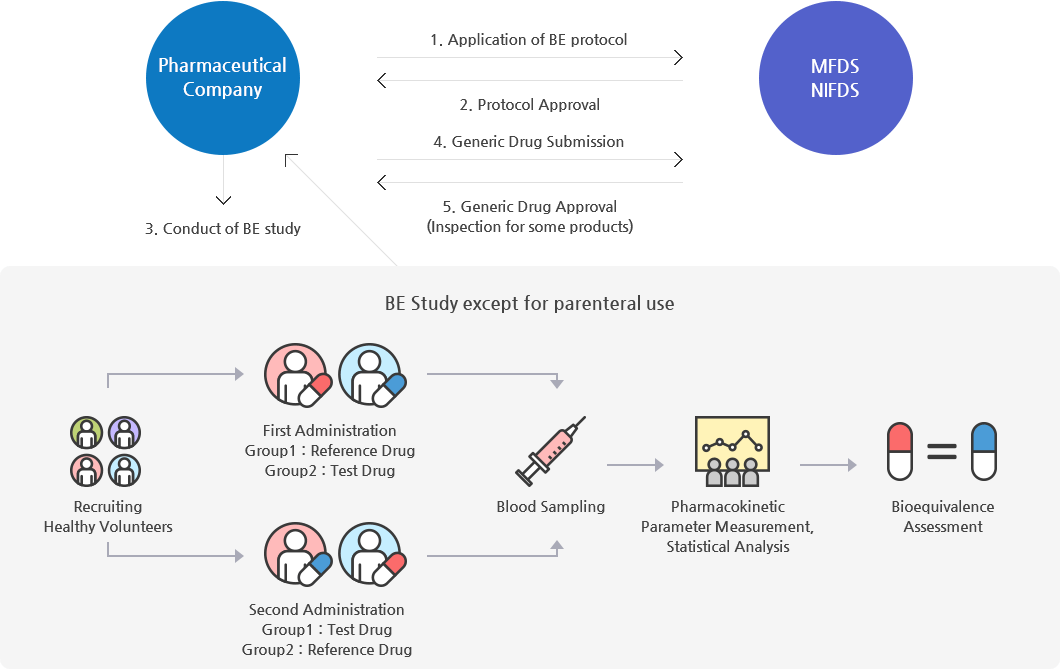
Pharmaceutical Company and MFDS,NIFDS interact as shown below.
1.Application of BE protocol
2.Protocol Approval
4.Generic Drug Submission
5.Generic Drug Approval(Inspection for some products)
Pharmaceutical Company influences 3.Conduct of study
Be Study except for parenteral use influences Pharmaceutical Company
Be Study except for parenteral use is Recruiting Healthy Volunteers -> First Administration(Group1:Reference Drug Group2:TestDrug), Second Administration(Group1:TestDrug Group2:Reference Drug)
-> Blood Sampling -> Pharmacokinetic Parameter Measurement, statistical Analysis -> Bioequivalence Assessment
Zoom
Pharmaceutical Company and MFDS,NIFDS interact as shown below.
1.Application of BE protocol
2.Protocol Approval
4.Generic Drug Submission
5.Generic Drug Approval(Inspection for some products)
Pharmaceutical Company influences 3.Conduct of study
Be Study except for parenteral use influences Pharmaceutical Company
Be Study except for parenteral use is Recruiting Healthy Volunteers -> First Administration(Group1:Reference Drug Group2:TestDrug), Second Administration(Group1:TestDrug Group2:Reference Drug)
-> Blood Sampling -> Pharmacokinetic Parameter Measurement, statistical Analysis -> Bioequivalence Assessment
Figure 1. the Process of Generic Drugs Approval
- RFDA approve generic drugs via drug approval evaluation
- In the whole process of drug review and approval, consult with Central Pharmaceutical Affairs Advisory Committee (CPAC), if necessary.